📞  — free, 24/7
— free, 24/7
 — free, 24/7
— free, 24/7
| Dosage | Package | Price per Dose | Price | |
|---|---|---|---|---|
| 5mg | 120 pills | C$4.02 | C$568.34 C$483.09 Best Price Popular | |
| 5mg | 90 pills | C$4.12 | C$436.48 C$371.01 | |
| 5mg | 60 pills | C$4.44 | C$313.71 C$266.66 | |
| 5mg | 30 pills | C$5.08 | C$179.58 C$152.64 |
Disclaimer: This page provides general information about fincar. It is intended for educational use and does not replace medical advice from a healthcare professional. Consult a doctor or pharmacist for advice tailored to your situation.
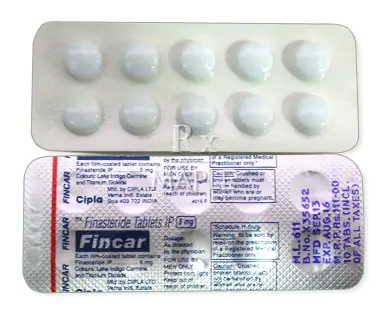
Fincar is a brand name for finasteride, a medicine used in men to treat conditions related to the prostate and to hair loss. Availability and access may vary by location and local regulations. A prescription may be required depending on local rules.
Finasteride works by interfering with the body’s processing of testosterone. It reduces the formation of a hormone called dihydrotestosterone (DHT), which is linked to prostate growth and certain types of hair loss in men. By lowering DHT levels, fincar can help with the symptoms of an enlarged prostate and may slow hair loss in some men.
Fincar tablets are taken by mouth and are typically prescribed in a specific strength for the condition being treated. The exact dose, duration, and whether it is the right medicine for you depend on medical history and your healthcare provider’s assessment. Always use as prescribed and do not switch brands or strengths without professional guidance.
Important safety notes: fincar is intended for use by men. It is not approved for use in women or children. People who are or may be pregnant should avoid handling broken or crushed tablets because of potential risk to a developing fetus. If accidental exposure occurs, seek medical advice promptly.
In general, fincar belongs to a class of medicines called 5-alpha-reductase inhibitors. It is used to treat symptoms of an enlarged prostate (benign prostatic hyperplasia, BPH) in men. By reducing DHT levels, it can help shrink the prostate and relieve urinary symptoms in some men.
Fincar may also be used as part of a broader approach to male pattern hair loss in some settings, depending on local labeling and available strengths. The medicine does not cure hair loss, but it may help slow further loss in certain cases while ongoing treatment continues to be used under medical supervision.
Not every person with BPH or hair loss will respond the same way. Some individuals may experience improvement, while others may notice limited or no benefit. The decision to use fincar should be made in consultation with a healthcare professional who can weigh benefits against possible risks.
In Canada and other jurisdictions, the exact approved uses, dosing, and product labeling may vary. Always follow the guidance provided by the prescribing clinician and the product information leaflet. If any use is unclear, consult a pharmacist for clarification before starting therapy.
Finasteride, the active ingredient in fincar, works by inhibiting the enzyme that converts testosterone into dihydrotestosterone (DHT). Lower DHT levels can lead to a reduction in prostate size and a slowing of hair loss progression in men who have those conditions.
The effect on urinary symptoms from BPH or on hair loss often takes time to become noticeable. Some people see improvements after several weeks, while others may require many months of continued therapy. Responses vary and are influenced by individual factors and adherence to treatment.
Finasteride does not cure BPH or male pattern hair loss. If treatment is stopped, symptoms may return or progress. Long-term treatment decisions should be made with a healthcare professional who can monitor response and any side effects.
Fincar interacts with the hormonal balance in the body in ways that may affect other medicines and tests. Before starting therapy, discuss all medical conditions and medications with a clinician to ensure it is appropriate for the individual situation. If symptoms change unexpectedly, contact a healthcare professional for guidance.
Use fincar exactly as prescribed by a healthcare professional. Do not modify the dose or frequency without medical advice. If there is any doubt about how to take the medicine, ask a pharmacist for instructions.
Take the tablet by mouth with water. It can be taken with or without food, depending on what your clinician recommends. Do not split, crush, or chew the tablet unless instructed by a healthcare provider. Swallow the tablet whole for best absorption and effect.
If a dose is missed, resume your normal dosing schedule as directed by your clinician. Do not take a double dose to make up for a forgotten one. If you are unsure what to do after a missed dose, contact a pharmacist or doctor for guidance.
Store fincar at room temperature, away from moisture, heat, and direct light. Keep it out of reach of children and pets. If you have questions about storage or shelf life, ask a pharmacist for specifics. Always discard medicines according to local guidelines when they are no longer needed or have expired.
Fincar is intended for use by men for whom it has been prescribed. It is not for women or children. Pregnant people should avoid handling broken or crushed tablets due to potential risk to a male fetus; if exposure occurs, seek medical advice promptly.
People with certain liver conditions or other medical problems should discuss fincar with a healthcare provider before starting treatment. Some medicines may interact with fincar, or fincar may interact with other treatments, so a full medication review is important.
Tell your clinician about all medicines you currently take, including over-the-counter products, vitamins, and herbal supplements. Do not start new medicines without professional advice, and do not discontinue prescribed therapy without a clinician’s guidance.
If you have a history of breast disease or not certain about safety in special populations (such as older adults), discuss these concerns with a clinician. A careful assessment helps determine if fincar is appropriate and safe for the individual’s health profile.
All medicines carry potential risks. A clinician will weigh the potential benefits of fincar against possible side effects or interactions for the individual patient. If new symptoms arise, contact a healthcare professional.
Allergic reactions to fincar are possible, though rare. Seek urgent medical help if there are signs of a severe allergic reaction, such as swelling of the face or throat, severe rash, or trouble breathing.
Some people may notice changes in their breast tissue, such as tenderness, swelling, or discharge. If breast changes occur, inform a clinician promptly, as this may require evaluation.
Fincar may affect certain laboratory tests, including PSA tests used in evaluating prostate health. If a PSA test is planned, inform the healthcare provider that finasteride is being used, as interpretation may be affected. This is important for accurate assessment by clinicians.
Common side effects may include a decrease in sexual desire, difficulty with sexual function, or changes in ejaculate. These effects are reported by some individuals taking finasteride and may improve with time or after stopping treatment.
Less common side effects can include breast tenderness or enlargement, skin rash, or dizziness. If any new or unusual symptoms appear, contact a healthcare professional for evaluation.
In rare cases, people may experience mood changes, depression, or thoughts of self-harm. If mood changes occur, seek medical advice promptly.
More serious side effects requiring immediate medical attention include signs of an allergic reaction, or a breast lump or nipple discharge that is not typical for the person. If such symptoms occur, seek medical help urgently.
Fincar may interact with other medicines. A clinician will review all medications to minimize risks. Do not start or stop any medicine without consulting a healthcare professional.
Some interactions may require dose adjustments or closer monitoring. It is important to inform the doctor about all current treatments, including over-the-counter medicines, vitamins, and herbal products.
Finasteride can affect the results of certain laboratory tests. Mention its use to the testing clinician so interpretation of results can be accurate. If a PSA or other test is planned, discuss potential implications with a healthcare provider.
Certain combinations of medicines or conditions may increase the risk of side effects. If you notice unusual symptoms after starting fincar, contact a clinician promptly for guidance. Do not make changes without advice from a professional.
Finasteride can pose risks if a pregnant person is exposed to the medication. Pregnant individuals should avoid handling tablets that are broken or crushed due to potential risk to a developing male fetus. If exposure occurs, seek medical advice promptly.
This medicine is intended for use in men and is not suitable for use by women who are pregnant or might become pregnant. If a partner is pregnant and there is a possibility of contact with semen or other bodily secretions, discuss precautions with a clinician.
Always store the medication securely to prevent accidental exposure. If pregnancy is planned in the future or there is any chance of pregnancy, talk with a healthcare professional about risks and alternatives.
For anyone who is pregnant or may become pregnant, it is important to discuss all treatment options with a healthcare provider to determine safe and effective care. Do not rely on general advice for a pregnancy decision; seek individualized medical guidance.
Improvements in urinary symptoms may appear after some weeks of treatment, but it can take several weeks to months for meaningful effects. Individual responses vary, and ongoing evaluation by a clinician helps determine whether the treatment is effective for the person.
For hair-related use, changes may take several months to become noticeable. Not all individuals will see the same level of improvement, and results may depend on factors such as genetics and disease progression. A clinician can help set expectations.
If a dose is missed, resume your normal dosing schedule as advised by your clinician. Do not double the dose to make up for the missed one unless instructed by a health professional.
Combination use with other hair loss therapies should be guided by a clinician. Some products may interact or affect effectiveness. Always discuss new or ongoing treatments with a healthcare professional before adding them.
Moderate or occasional alcohol use is not generally prohibited by fincar prescribing information, but alcohol can affect overall health and symptom perception. If there are concerns, discuss them with a clinician.
Finasteride has been used in adults of various ages. In older adults, clinicians consider overall health, potential interactions with other medicines, and existing medical conditions before continuing therapy. Ongoing monitoring is important.
Most people can drive or operate machinery while taking fincar, provided they do not experience dizziness or other side effects. If any side effects occur that could impair judgment or reaction time, avoid driving and seek medical guidance.
Stoppage should be discussed with a healthcare professional. Some benefits may wane after stopping, and restarting treatment should follow medical advice to ensure safety and effectiveness.
Breast changes or lumps warrant medical evaluation. Although rare, these symptoms may require a clinician’s assessment to rule out other conditions. Seek medical advice promptly if new breast symptoms appear.
Finasteride can influence PSA measurements used in prostate cancer screening. If a PSA test is planned, inform the clinician that fincar is being used so interpretation can be adjusted accordingly. Do not rely on PSA results without professional guidance.
Ask a pharmacist or healthcare professional for product labeling, patient information leaflets, and official guidance in your region. They can provide personalized information tailored to medical history and current health needs.
Official product labeling and patient information leaflets for finasteride as used in Canada and other regions.
Health Canada drug information resources and safety notices related to finasteride products.
Pharmacist and clinician guidelines used in routine medical practice for the management of benign prostatic hyperplasia and androgen-related hair loss in men.
14–21 days. Free from C$276.06 .
5–9 days. C$41.41
−10% when paying with cryptocurrency.
−10% on all repeat orders.
All orders are packed in neutral, unbranded boxes with no product name on the outside.
